Answers
Answer:
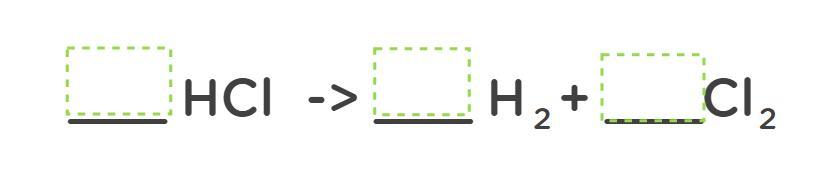
2HCl - H2 + Cl2
Explanation:
The two hydrochloric acid molecules decompose into one hydrogen molecule (H2) and one chlorine molecule (Cl2). If there is only one type of a specific molecule present, you should not have to add a coefficient of 1 behind it.
Related Questions
An object's velocity can be described by its acceleration and direction.
True
False
Answers
Answer:
False
Explanation:
An object's velocity can be described by it's direction. Because velocity is a vector. Besides velocity and acceleration are different units they can't be described by each other.
how many moles are in 3.4 x 10^23 molecules of carbon dioxide CO2
Answers
3.4x10^23/6.02x10^23 = .565
Rounded to the thousandths
HELP ASAP - How much energy is lost when 80.0 grams of steam at 100.C turns into liquid water at 100.C?
Answers
Answer:
Q = 43,000 calories = 180,000 joules = 180 Kilojoules
Explanation:
Heat flow for phase change can be defined as Q = m·ΔHv
ΔHv = Heat of Vaporization for water = 540 calories/gram = 2259 joules/gram = 2.259 Kilojoules/gram
m = mass of object of interest ( in this problem 80 grams steam 100°C converting to 80 grams of water at 100°C
Q = m·ΔHv = 80g x 540 cals/g =43.200 calories x 4.184 joules/cal = 180,748.8 joules (calculator answer) => 180,000 joules (2 sig. figs.) = 180 Kj
the first plants to grow after a fire are called?
Answers
Answer:
Ephemerals
Explanation:
The first plants to move into the new bare ground after a wildfire are wildflowers or “weeds.” These fast-germinating, leafy herbaceous plants are also known as “forbs” or “ephemerals.” They quickly germinate, grow and produce a new crop of seeds.
Answer:
ephemeral
Explanation:
In addition to having a blue color what other characteristic do Neptune and Uranus share??
no rings
27 moons
no axis tilt
cold temperatures. HELP ASP
Answers
Answer:
d
Explanation:
it just makes sense
Answer:
Cold temperatures!
Explanation:
Both planets are very far from the sun, now to explain how the other answers would fit or just dont make sense, No rings, the answer isn't the best fitting answer for this question, 27 moons, this is wrong because neptune has 14 moons, only uranus has 27. No axis tilt, this answer is wrong because both planets have an axis.
7. DNA is essential to living organisms because it O is a spiral helix can prevent disease O carries the genetic code O can break apart
Answers
Answer: DNA is essential...because it carries the genetic code.
Explanation:
Although the other statements are true about DNA, it's a spiral helix and can break apart, its main function is that it contains the genetic code for organisms.
Can you help give examples of elements, compounds, and mixtures? My friend is having trouble with it.
Answers
Answer:
Elements are all the subtances on the periodic table. Compound are elements that are fused by chemical means like potassium chloride. Mixture are elements that are not chemically fused but just in a sense mixed together.
PLS HELP!!! Which of the following is NOT a basic assumption of the Kinetic Molecular Theory?
a. The size of the particles is negligibly small
b. The kinetic energy is proportional to the temperature
C. Collisions of particles is elastic
d. The quantity of particles is negligible
Answers
Answer:
it might be the second answer
Which of the following is considered a compound?
1) N2
2) Na
3) BF3
Answers
A compound is composed of 2 or more different elements.
Na is only 1 element- sodium
N2 is 2 of the same element- 2 nitrogen elements
what is the chemical equation for this?
Answers
Answer:
J
Explanation:
Chlorine Gas is Cl2 so if it isnt on the left as that its wrong.
Then on the products see that Fe III means Fe is +3 charge so we have to have 3 Cl to balance it to 0 since each cl as -1 charge.
FInally check if the equation is balanced J is maybe H but its to blurry to tell
What is the mass number of a sulfur (S) atom that has 17 neutrons?
Answers
How many phosphorus atoms are in two moles of the compound Mg3(PO4)2? O 4 atoms O 1.2 x 1023 O 2 atoms O 2.4 x 1023
Answers
The number of phosphorus atoms : 2.408 x 10²⁴
Further explanationThe mole is the number of particles(molecules, atoms, ions) contained in a substance
1 mol = 6.02.10²³ particles
Can be formulated
N=n x No
N = number of particles
n = mol
No = Avogadro's = 6.02.10²³
moles of Mg₃(PO₄)₂ = 2
moles of phosphorous(P) atoms in Mg₃(PO₄)₂ ⇒ mol ratio of elements in the compound(for P=2)
[tex]\tt 2\times 2=4[/tex]
The number of atoms of phosphorous(P) :
[tex]\tt 4\times 6.02\times 10^{23}=2.408\times 10^{24}[/tex]
A student prepared this table to gather information about digestion. Digestion of Food Column A Column B Physical Changes Chemical Changes The results of which investigation would be entered in Column A of the table? A. How do mitochondria release the energy in food molecules? B. Which type of teeth are most important in crushing food? C. What chemicals in the stomach are most important in breaking down proteins? D How do substances released by the pancreas affect the rate of digestion?
Answers
Answer:
B. Which type of teeth are most important in crushing food?
Explanation:
According to the question physical changes ought to be entered into column A while chemical changes ought to be entered into column B.
The crushing of food is a physical change. It does not involve any chemical transformation of the food material.The other options involve some sort of chemical transformation taking place on the food material being digested.
A student wants to find out if drinking coffee will affect the amount of sleep she has everyday. she collected data of cups of coffee that she drank to how many hours of sleep she got everyday. what would you plot on the x-axis?
Answers
We plot the cups of coffee drink on the X-axis, whereas hours of sleep are plotted on the Y-axis.
plotting graphs relationship between various sets of variables with respect to one another. the main use of a graph is it helps us to present data which are complex to describe in the text.Since in the problem it is given that, the student wants to record the hours of sleep which are affected by the number of cups of coffee consumed by her or him, therefore we will plot the number of cups of coffee on the X-axis and the number of hours of sleep will be plotted in Y-axis .
To learn more about Plotting graphs refer to the link https://brainly.com/question/3638574?referrer=searchResults.
#SPJ4
All atoms try to achieve---------------(minimum energy,stable energy,maximum energy,covalent energy)
Answers
Answer:
minimum energy
Explanation:
minimum energy means maximum stability
please mark me brainliest
PLEASE ANSWER “Tessa uses a toy slingshot to launch a tennis ball across the park for her dog to fetch. For her first launch, she
uses 100 N of force. Her second launch uses 200 N of force, and her third launch uses 300 N. Which launch had
the greatest acceleration of the tennis ball?”
Answers
The third launch ( with 300 N force) had the greatest acceleration of the tennis ball
Further explanationNewton's 2nd law explains that the acceleration produced by the resultant force on an object is proportional and in line with the resultant force and inversely proportional to the mass of the object
∑F = m. a
[tex]\large{\boxed{\bold{a=\frac{\sum F}{m}}}[/tex]
F = force, N
m = mass = kg
a = acceleration due to gravity, m / s²
From the above equation it has been shown that the force acting on the object is directly proportional to its acceleration, so the greater the force exerted on the object, the greater the acceleration of the object produced.
what is 2100 ÷ 35399 pleasss help me
Answers
Answer:
0.0593237097
Explanation:
Anything helps thank you
Answers
3 x 2 because double the C3N
Calcium carbonate forms limestone, one of the most common rocks on Earth. It also forms stalactites, stalagmites, and many other types of formations found in caves. When calcium carbonate (CaCO3) is heated, it decomposes to form solid calcium oxide and carbon dioxide gas. Write the balanced equation and calculate how many liters of carbon dioxide will be produced at STP if 2.38 g of calcium carbonate reacts completely?
Answers
Answer: [tex]CaCO_3(s)\rightarrow CaO(s)+CO_2(g)[/tex] , 0.533 L
Explanation:
According to the law of conservation of mass, mass can neither be created nor be destroyed. Thus the mass of products has to be equal to the mass of reactants. The number of atoms of each element has to be same on reactant and product side. Thus chemical equations are balanced.
[tex]CaCO_3(s)\rightarrow CaO(s)+CO_2(g)[/tex]
According to avogadro's law, 1 mole of every substance occupies 22.4 L of volume at STP.
Number of moles =[tex]\frac{\text {given mass}}{\text {Molar mass}}[/tex]
moles of calcium carbonate = [tex]\frac{2.38g}{100g/mol}=0.0238mol[/tex]
As 1 mole of calcium carbonate produces = 22.4 L of carbon dioxide at STP
Thus 0.0238 moles of calcium carbonate produces =[tex]\frac{22.4 L}{1}\times 0.0238=0.533L[/tex] of carbon dioxide at STP
The balanced equation is [tex]CaCO_3(s) \rightarrow CaO(s) + CO_2(g)[/tex]
The number of liters of carbon dioxide that should be produced is 0.533L
Law of conversation of mass:As per this law, the mass could neither be developed nor be destroyed. So, the mass of products should be equivalent to the mass of reactants. The number of atoms of every element has to be the similar on reactant and product sides. Due to this, the chemical equations are balanced.
[tex]CaCO_3(s) \rightarrow CaO(s) + CO_2(g)[/tex]
Avogadro's law:As per this law, 1 mole of every substance occupies 22.4 L of volume at STP.
So,
We know that
Number of moles = [tex]given\ mass \div molar\ mass[/tex]
Now the moles of calcium carbonate is [tex]= 2.38 \div 100[/tex] = 0.0238 mol
Since 1 mole of calcium carbonate generated = 22.4 L of carbon dioxide at STP
So, 0.0238 moles of calcium carbonate generated
[tex]= 22.4 \div 1 \times 0.0238 \\\\= 0.533L[/tex]
of carbon dioxide at STP
Learn more about the equation here: https://brainly.com/question/15844891
A solution of silver chloride reacts with aqueous sodium sulfate to produce a precipitate and a solution.
When balanced, the coefficients for this chemical reaction, is?
Answers
Answer:
<br> a) Barium chloride and sodium sulphate aqueous solutions react to give insoluble barium sulphate and aqueous solution of sodium chloride.
How does one balance an equation?
Answers
Answer:
To make both sides of an equation equal, multiply the number of atoms in each element until both sides are equal.
Explanation:
The chemical equation is an equation in which the number of atoms of each element on the product side is equal to the number of atoms of each element on the reactant side. To make both sides of an equation equal, multiply the number of atoms in each of the elements on both the sides until both the sides become equal.
In testing for the presence of lead 2+ ions in solution, a student addes a
solution of sodium nitrate. What will be the conclusion of the test if no
precipitate forms? *
Answers
is it true that Einstein's equation E=mc2 states that matter can be converted to energy. This is an exception to the Law of Conservation of Mass.
Answers
The famous formula E=mc^2 really says that mass and energy are the same thing, but measured in different units. So the separate laws of conservation- one for mass and the other for energy- are now merged into one law.
Einstein's theory of special relativity (1905) shows that matter (as mass) and energy can be converted into each other according to the famous equation E = mc2, where E is energy, m is mass, and c is the speed of light.
Hope It helps you.
3. Which term is defined as "anything that has mass and occuples space"?
Answers
Answer:
matter
Explanation:
All physical 'stuff'
Answer:
matter
Explanation:
Matter is anything that has mass and occupies space .
The pressure on 2.50 L of gas is changed from 4.5 atm to 2.2 atm. What is the new volume of gas?
Answers
Answer:
5.11 LExplanation:
The new volume can be found by using the formula for Boyle's law which is
[tex]P_1V_1 = P_2V_2[/tex]
Since we are finding the new volume
[tex]V_2 = \frac{P_1V_1}{P_2} \\[/tex]
From the question we have
[tex]V_2 = \frac{2.5 \times 4.5}{2.2} = \frac{11.25}{2.2} \\ = 5.1136666...[/tex]
We have the final answer as
5.11 LHope this helps you
What general trend do you observe in the periodic table of the Atomic Radius?
Answers
Answer:
Atomic radius Increases in groups from top to bottom and decrease in period from left to right.
Explanation:
how many Nitrogen are in 2AlN
Answers
If you have 250 mL of 0.75 M LINO2.
How many grams of this substance
would you have?
Answers
Mass of this substance = 9.928 g
Further explanationMolarity is a way to express the concentration of the solution
Molarity shows the number of moles of solute in every 1 liter of solute or mmol in each ml of solution
[tex]\large{\boxed {\bold {M ~ = ~ \frac {n} {V}}}[/tex]
Where
M = Molarity
n = Number of moles of solute
V = Volume of solution
So to find the number of moles can be expressed as
[tex]\boxed{\bold{n=V\times M}}[/tex]
mol of substance -Lithium nitrite - LiNO₂ :
V = 250 ml = 0.25 L
M = 0.75 M
[tex]\tt n=0.25\times 0.75=0.1875[/tex]
So mass of this substance - LiNO₂ (MW=52,947 g/mol) :
[tex]\tt mass=mol\times MW\\\\mass=0.1875\times 52,947 g/mol\\\\mass=9.928~g[/tex]
What relationship do you observe between the temperature and density?
Answers
Answer:Density is directly proportional to pressure and indirectly proportional to temperature. As pressure increases, with temperature constant, density increases. Conversely when temperature increases, with pressure constant, density decreases.
Explanation:
hope this helped
Calculate the molar mass of a gas with a mass of 3.15 g, a pressure of 785 mmHg, and a temperature of 20 Celsius. The gas has a volume of 750 ml.
Answers
Answer:
Molar mass = 105 g/mol
Explanation:
Given data:
Mass of gas = 3.15 g
Pressure = 785 mmHg
Temperature = 20°C
Volume of gas = 750 mL
Molar mass of gas = ?
Solution:
Pressure = 785 mmHg (785/760 = 1 atm)
Temperature = 20°C (20+273 = 293 K)
Volume of gas = 750 mL (750 mL× 1 L/1000 mL = 0.75 L)
PV = nRT
R = general gas constant (0.0821 atm.L/mol.K)
1 atm× 0.75 L = n×0.0821 atm.L/mol.K × 293 K)
0.75 atm.L = n× 24.06 atm.L/mol
n = 0.75 atm.L / 24.06 atm.L/mol
n = 0.03 mol
Molar mass of gas:
Molar mass = Mass / number of moles
Molar mass = 3.15 g/ 0.03 mol
Molar mass = 105 g/mol